
SARS-CoV-2 review featured on cover of Nature Reviews Molecular Cell Biology
The review focuses on the cellular and structural foundations of viral entry mechanisms.

Virologist focused on gene therapy and SARS-CoV-2. Trained in the labs of Hyeryun Choe and Mike Farzan at Scripps Research and Harvard Medical School/Boston Children's Hospital . MD at Florida Atlantic University . Internal Medicine Resident at OHSU .
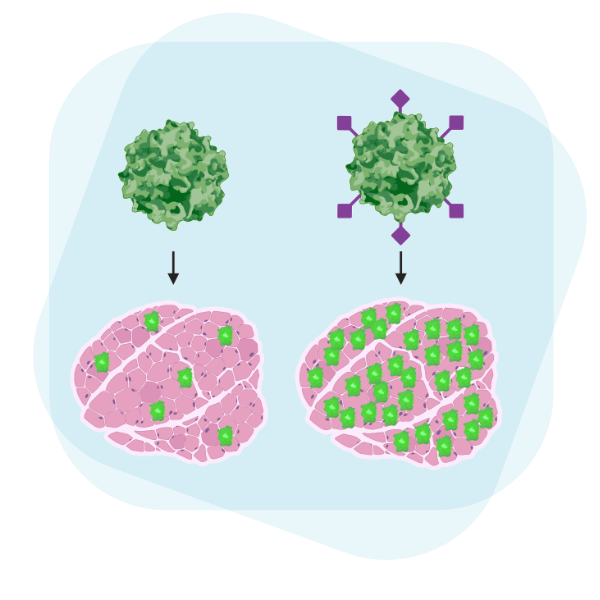
Skeletal muscle is an ideal gene therapy target to act as a factory for the production and secretion of systemically-acting therapeutic proteins. However, current gene delivery technologies are limited in transduction efficiency in skeletal muscle. We developed a novel AAV vector targeting insulin receptor to achieve enhanced transduction of skeletal muscle in vivo with a modular approach that can be combined with other advances in AAV capsid engineering.
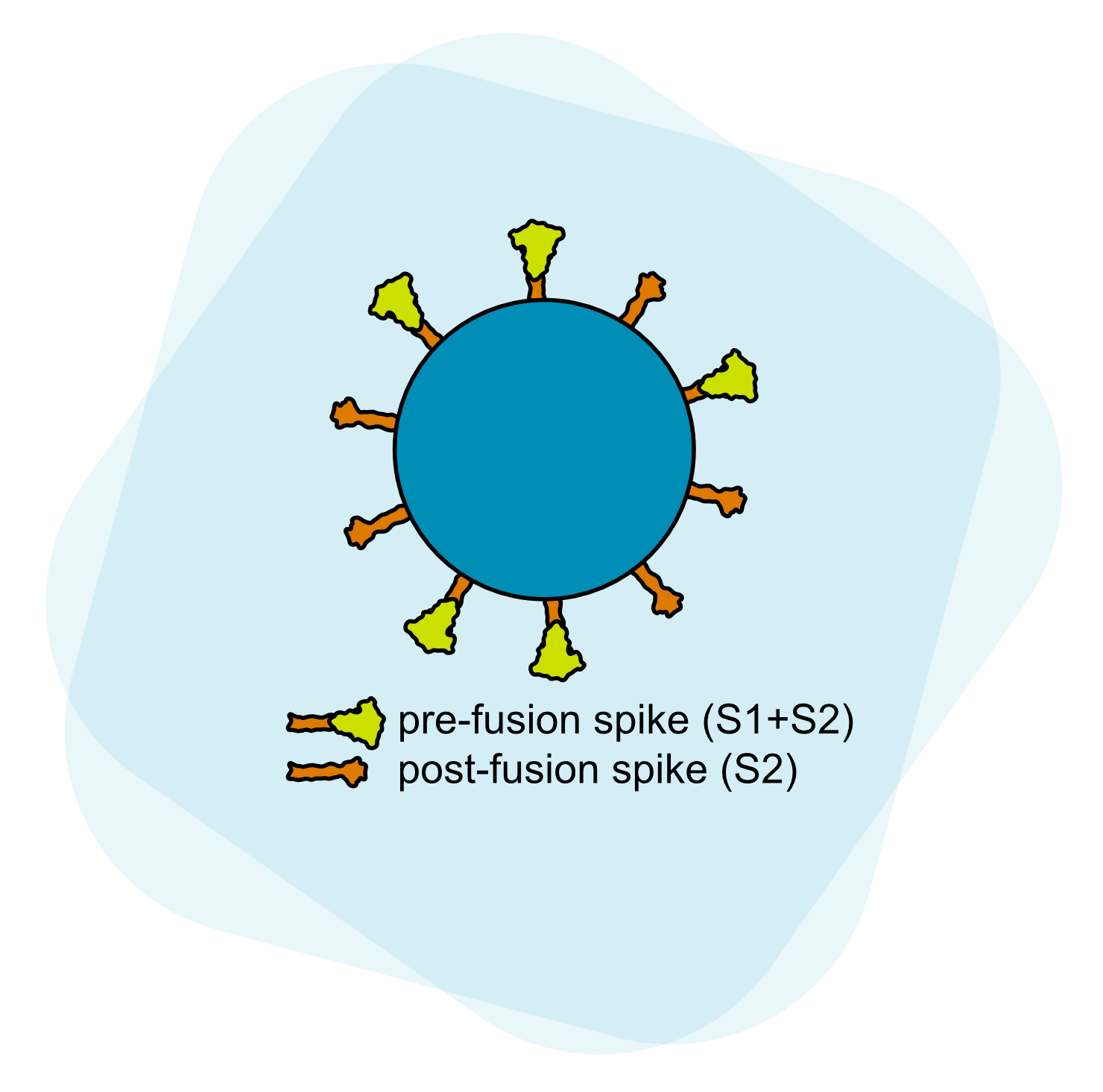
SARS-CoV-2, the causative agent of COVID-19, binds and enters host cells by action of its spike protein. SARS-CoV-2, unlike other sarbecoviruses, has acquired a multibasic cleavage site at the junction of the S1 and S2 subunits which is processed by furin-like proprotein convertases in the virus-producer cell before encountering target cells. Therefore, intermolecular association of S1 and S2 subunits are necessary to retain infectivity. Our work has clarified the nature of this interaction and revealed the mechanism of increased infectivity of the D614G variant by the effects of this amino acid change on S1-S2 interaction and spike protein density.
For more information, please see my full list of publications. For related projects, please visit the website of Dr. Hyeryun Choe.

The review focuses on the cellular and structural foundations of viral entry mechanisms.

Work by Jackson et al. demonstrates greatly enhanced gene delivery to skeletal muscle by adeno-associated virus vectors modified to target insulin receptor.

The dissertation is focused on the engineering of a novel gene therapy vector for transduction of skeletal muscle.